
Our
Story
Get to Know Us
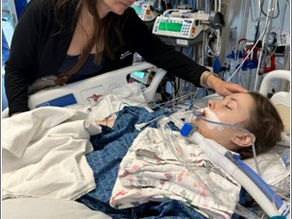
At Transplant Families, our mission is to unite families navigating the complexities of pediatric solid organ and bone marrow transplants. We offer inspiration, support, and education, empowering patients and their families to embrace life fully – before, during, and long after transplant.
Save Your Spot
No events at the moment
News

To reach the Platinum level, Transplant Families added extensive information to its Nonprofit Profile on GuideStar: basic contact and organizational information; in-depth financial information; qualitative information about goals, strategies, and capabilities; and quantitative information about results and progress toward its mission. By taking the time to provide this information, Transplant Families has demonstrated its commitment to transparency and to giving donors and funders meaningful data to evaluate Transplant Families.








_edited.png)

